

AD109: A Novel Neuromuscular Approach In Development to Improve Oxygenation and Address Unmet Need
If approved, we believe AD109 could be the first oral pharmacological treatment to improve oxygenation during sleep by directly addressing the underlying neuromuscular cause of upper airway collapse in people with obstructive sleep apnea (OSA). No mask or device would be needed—the potential power of AD109 has been developed in a pill.
The Synergistic Science of AD109
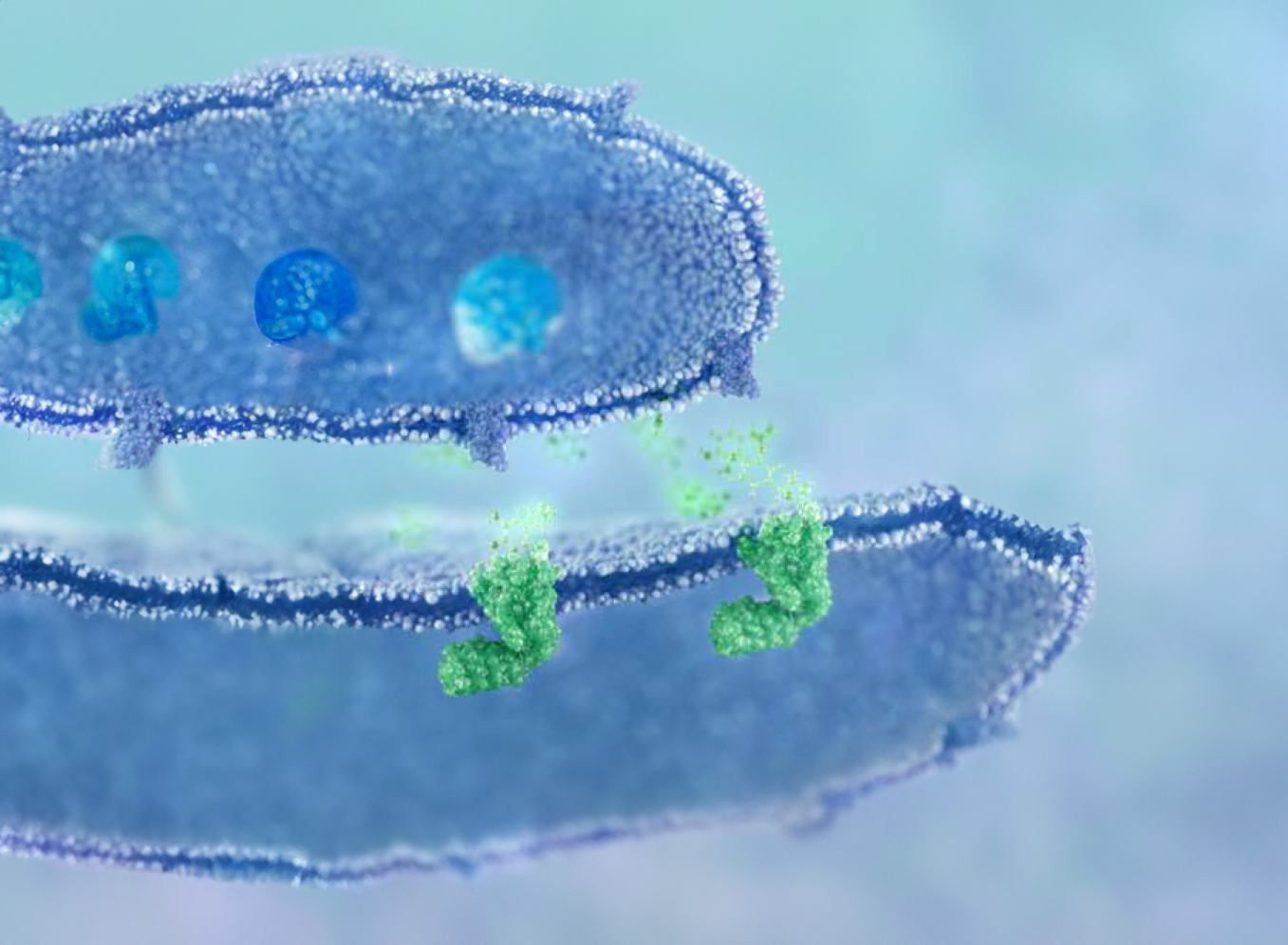
AD109 is an investigational first-in-class, fix-dose oral combination of aroxybutynin, a novel antimuscarinic, and atomoxetine, a selective norepinephrine reuptake inhibitor.

By targeting the underlying neuromuscular dysfunction of OSA, AD109 offers an innovative therapy that has the potential to improve the lives of millions of people living with this serious chronic disease.
Hoyee Leong
PhD, Vice President, Medical Affairs

A Potential Solution
for Oxygenation
We believe AD109 represents potential new hope for people living with OSA who have either refused, abandoned, or are currently dissatisfied with their treatment.
The investigational therapy is a once-at-bedtime oral pill that lowers the complexity of intervention and may help more people benefit from effective, restorative sleep.
AD109 Clinical Program

Visit LunAIRo on clinicaltrials.gov
View LunAIRo Phase 3 Topline Results

Apnimed has completed SynAIRgy, a Phase 3, randomized, double-blind, placebo-controlled, 6-month parallel-arm clinical trial that evaluated AD109 vs placebo in participants with mild to severe OSA. Topline results of this trial were announced on May 19, 2025, highlighting that AD109 met the primary endpoint, mean change in apnea-hypopnea index (AHI4, p<0.0001) at 26 weeks, across a wide range of weight classes enrolled and a broad range of people with mild, moderate and severe obstructive sleep apnea.
Visit SynAIRgy on clinicaltrials.gov
View SynAIRgy Phase 3 Topline Results

Apnimed has completed MARIPOSA, a Phase 2b randomized, double-blind, placebo-controlled, parallel-arm, dose-finding clinical trial that evaluated AD109 as a possible treatment for OSA in a broad range of OSA patients over a 1-month period. The trial compared fixed-dose combinations of AD109 to placebo and atomoxetine to placebo. The study results showed that AD109 achieved a statistically significant reduction of AHI4 compared to placebo (P=0.001 vs placebo). AHI4 was decreased by AD109 across the full duration of the night, demonstrating that clinical activity was present across the beginning, middle, and end of the sleep period. Dosing with AD109 led to clinically important reductions in AHI in most patients with mild, moderate, and severe OSA. MARIPOSA results also showed that AD109 improved daytime fatigue, an often-debilitating side effect of OSA.
For Additional Information:


Stay Updated
Submit your email for Apnimed company news, including updates on AD109.


Explore Our Full Pipeline
AD109 is only the beginning.
